What is the Lewis structure for C2H2Cl2?
Is C2H2Cl2 polar or nonpolar?
Question: For C2H2Cl2, if arranged with the two H next to each other and the two Cl next to each other (in cis arrangement), it results in a
polar molecule.
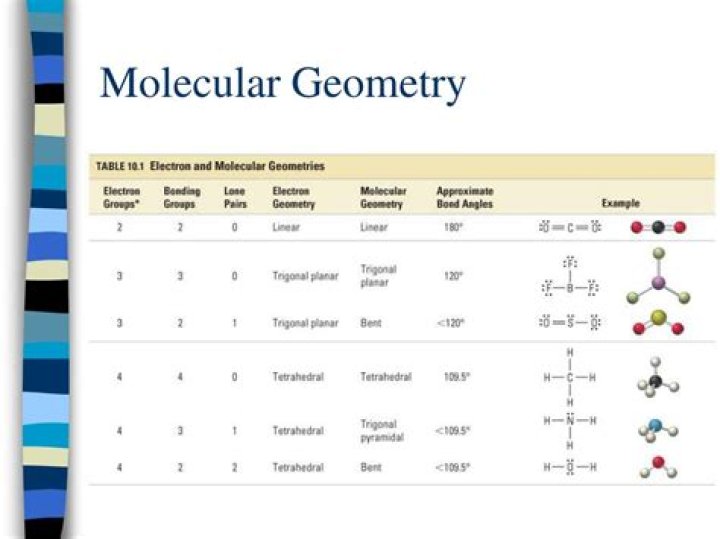
What is the molecular geometry for CH2O?
Formaldehyde (CH2O) lewis dot structure, molecular geometry, polar or non-polar, hybridization
| Name of Molecule | Formaldehyde |
|---|
| Chemical formula | CH2O |
| Molecular geometry of CH2O | Trigonal planar |
| Electron geometry of CH2O | Trigonal planar |
| Hybridization | Sp2 |
Does C2H2Cl2 have a double bond?
With C
2H
2Cl2 there are only single bonds. Carbon is the least electonegative atom so it goes at the center of the C
2H
2Cl
2 Lewis structure. Remember that Hydrogen (H) atoms always go on the outside of a Lewis Structure. Note that Hydrogen only needs two valence electrons to have a full outer shell.
Is C2H2Cl2 tetrahedral?
Re: C2H2Cl2
The shape of C2H2Cl2is tetrahedral.
How many polar isomers are possible for C2H2Cl2?
Two isomers
Two isomers of C2H2Cl2 are polar.
How many double bonds does C2H2Cl2 have?
A chemical structure of a molecule includes the arrangement of atoms and the chemical bonds that hold the atoms together. The cis-1,2-Dichloroethene molecule contains a total of 5 bond(s) There are 3 non-H
bond(s), 1 multiple bond(s) and 1 double bond(s).
What is the hybridization of carbon in C2H2Cl2?
How does the hybridization of carbon change as it goes from C2H2 + Cl2 -> C2H2Cl2 ? The hybridization of carbon changes from sp to
sp3. There are 3 bonds in C2H2 molecule, however there are only two bonds connected to the central atom, C, therefore the hybridization would be sp.
Is C2H2Cl2 sp3 hybridized?
Question: Item 6 Part A Which of the following is true about the molecule of cis-C2H2Cl2?
The hybridization of both carbons is sp3. … The molecule is chiral. The H-C-Cl bond angle is approximately 120°.